

Reciprocal IVF is a treatment designed for female same-sex couples in which both partners are able to actively participate.


Unlike other fertility treatments, shared motherhood allows both partners to share a unique biological connection with their baby. The partner who provides the egg is connected genetically, while the other partner is connected through pregnancy, birth and breastfeeding.

Shared motherhood method involves an In-Vitro Fertilization (IVF) procedure, which has a higher success rate compared to Intra-uterine Insemination (IUI). The chances of IVF success with shared motherhood are also higher compared to the general population because couples who choose shared motherhood tend to be of a younger age and often do not face any fertility challenges. Our success rates are verified by the HFEA and updated annually. You can view them here.


The latest research in the field of epigenetics (the study of gene activity) suggest that the uterine environment plays a crucial role in embryonic development. Although the baby will inherit genetic characteristics from both the oocyte and donor sperm cell, he or she will also absorb nutrients directly from the birth mother, contributing the building blocks for the child’s development and growth. While the foetus remains in the amniotic fluid for nine months, the child’s gene expression is in fact determined by the prenatal environment of the uterus.

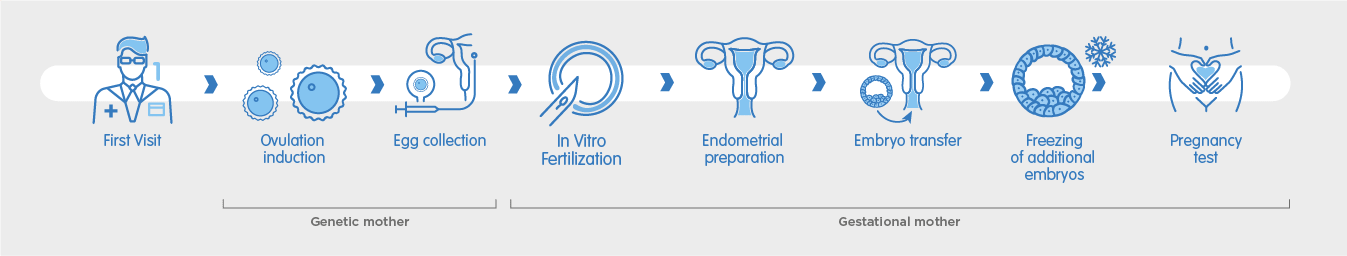
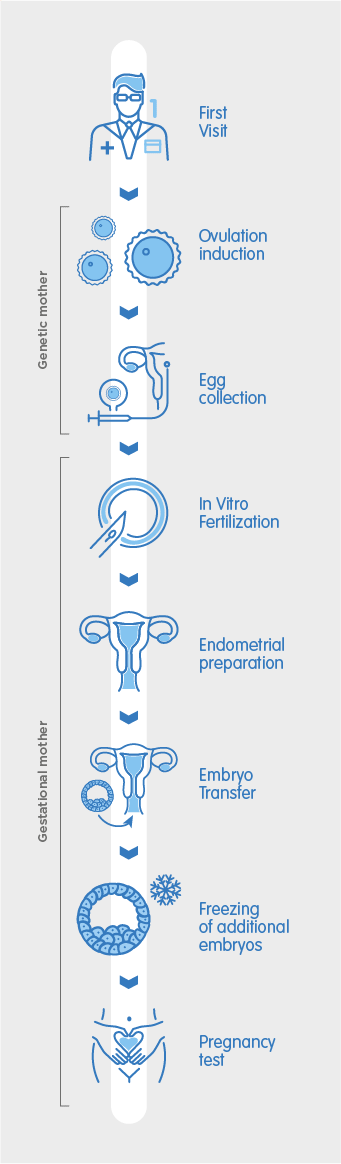


The first step to beginning your shared motherhood treatment is to visit our clinic for an initial consultation. During this consultation, our fertility specialists will discuss your medical history and perform ultrasound scans for both you and your partner in order to recommend the best way to maximise your chances of pregnancy.
The partner who will provide the egg begins ovarian stimulation, which consists of daily self-administered hormone injections over a period of between 10 – 20 days. Normally, a single egg is released each month. With hormonal stimulation, several eggs are able to develop, allowing multiple eggs to be collected without needing to repeat the ovarian stimulation process. When the oocytes have reached an appropriate number and size, a hCG injection is self-administered to trigger ovulation. The egg collection is scheduled 36 hours after the final trigger injection.



During the egg collection, the mature eggs are retrieved from the ovaries in a minor surgical procedure which takes place at the clinic. It is performed under sedation and should last no longer than 25 minutes. During the procedure, a cannula is inserted through the vagina to aspirate each of the ovarian follicles – the tiny sacs in which the eggs are located. Once collected, the eggs are taken to the laboratory to begin the IVF process.

The eggs are now fertilised in-vitro with the donor sperm inside the laboratory using one of two methods: conventional IVF or ICSI. Conventional IVF allows the egg and semen to mix in a culture dish where fertilisation can take place naturally, whereas ICSI is an assisted method which consists of injecting an individual sperm cell into each egg. Once fertilised, embryos are formed and incubated in the laboratory for 3 – 5 days, during which time they are monitored by our embryologists and classified according to their development and morphology. This classification system helps determine which embryo is best to transfer.



The innermost layer of the uterus, where the embryo implants, is called the endometrium. The partner who will receive the embryo and carry the pregnancy will begin a hormonal treatment in which oestrogens and progesterone are administered to prepare the endometrium for embryo transfer. This thickens the uterine lining until the correct endometrial thickness is achieved to maximise the chances of successful embryo implantation, and pregnancy.
Once it is established through an ultrasound scan that the correct endometrial thickness has been achieved, the healthiest embryo is transferred into the uterus through a thin cannula. This procedure is quick and painless, requiring no anaesthesia or aftercare. After the procedure, you can return home in no time.



If there are additional embryos of good quality remaining, they will be cryopreserved for future use. During vitrification, embryos are treated with a protective wash which prevents them from being harmed during the cooling process before being immersed in liquid nitrogen at a temperature of -196°C. They are then stored in special cryopreservation units. These units are equipped with a double alarm system to guarantee the temperature remains within at a very low temperature at all times. The frozen embryos can be used in a later cycle or to try for another baby without the need to undergo another cycle of stimulation.

In most cases, a blood pregnancy test can be performed 11 days after the embryo transfer. A blood test is more sensitive than a urine test, which can provide an unreliable result if performed too early. If the blood test result is positive, an ultrasound is scheduled within 20 days, after which you can be discharged and begin your antenatal care.

Reciprocal IVF, also known as Shared motherhood is an Assisted Reproduction treatment for same-sex female couples; this method allows both women to play a vital role in the conception, gestation and birth of their child.
The centre where you are having the treatment can provide donor sperm after selecting it according to rigorous genetic and immunological compatibility criteria.
The Shared motherhood procedure involves an IVF treatment, which has a higher success rate than insemination. The chances of success of IVF in Shared motherhood cases are also higher compared to the general population, because couples who choose this option are usually younger and often do not have any infertility problems.