

Pre-Implantation Genetic Testing for Aneuploidy (PGT-A), formerly known as Pre-Implantation Genetic Screening (PGS) can be combined with IVF treatment to detect chromosomal abnormalities in embryos. It is often recommended for specific patient groups who are at higher risk of miscarriage or have had recurring unsuccessful IVF cycles.


PGT-A (Preimplantation Genetic Testing for Aneuploidy), formerly known as Preimplantation Genetic Screening (PGS), is a genetic screening method performed on embryos during IVF treatment. PGT-A gives information about the genetic health of an embryo in order to help select the most suitable embryo for transfer. It can significantly improve the chances of implantation and reduce the risk of miscarriage. PGT-A is an area in which IVI specialise.
Today, aneuploidy screening is regularly offered to patients to give them the best possible chance of having a successful pregnancy and a healthy baby. Combining IVF and PGT-A treatment significantly increases the success rates per embryo transferred, following IVI’s “One Healthy Baby at a Time” philosophy.

Fewer cycles of IVF are needed.
By using PGT-A during IVF treatment, it multiplies the chances of success meaning fewer cycles are required to achieve a healthy pregnancy.
Reduced risk of miscarriage.
Chromosome aneuploidies are a primary cause of miscarriage. By transferring only embryos with balanced chromosomes, it significantly minimises the risk of miscarriage.
Increased chances of having a healthy baby.
For patients who have experienced recurring unsuccessful treatments, PGT-A can provide information about the genetic health of their embryos and indicate whether an alternative treatment might be required, such as egg donation.

RESULTS
Over the last 30 years, IVI has helped more than 250,000 dreams come true.
CARE
97% of our patients recommend IVI. We’re with you at every stage of your treatment, providing support and care.
TECHNOLOGY
IVI has a worldwide reputation for innovative research and has developed and patented pioneering techniques and technologies.
EXPERTISE
IVI is one of the largest fertility treatment providers in the world, with more than 75 clinics in 9 countries.
If PGT-A is recommended for your specific case, it will be explained in detail by your IVI consultant during your first consultation. You will then be able to make an informed decision before you begin your treatment cycle. IVF treatment with PGT-A will resemble the following pathway:


In a normal menstrual cycle, a woman grows a single follicle, producing a single egg (oocyte), each month. During IVF, the ovaries are stimulated to grow more follicles, producing more eggs. The aim of this is so that when the eggs are collected, there will be a higher cumulative chance of successful fertilisation within the laboratory and a higher chance of developing multiple good quality embryos that can be transferred back into the uterus.

When the follicles have reached an adequate size and number, it is time to schedule the oocyte retrieval procedure, known as an egg collection. This takes place around 36 hours after an hCG injection is administered, which induces ovulation. Your hCG injection is administered at home and you will be given clear instructions on how, and when, it needs to be administered. The egg collection takes place in the clinic under light sedation, so you won’t feel any discomfort during the procedure. It lasts approximately 15 minutes.

The process of fertilisation takes place in the laboratory. The insemination can be performed via conventional IVF, which involves placing an oocyte surrounded by spermatozoa in a culture plate, or via an intracytoplasmic sperm injection (ICSI), which involves depositing a live spermatozoon directly into the oocyte using a pipette. ICSI is only recommended in specific cases, and your consultant will explain this in detail if this is advised.
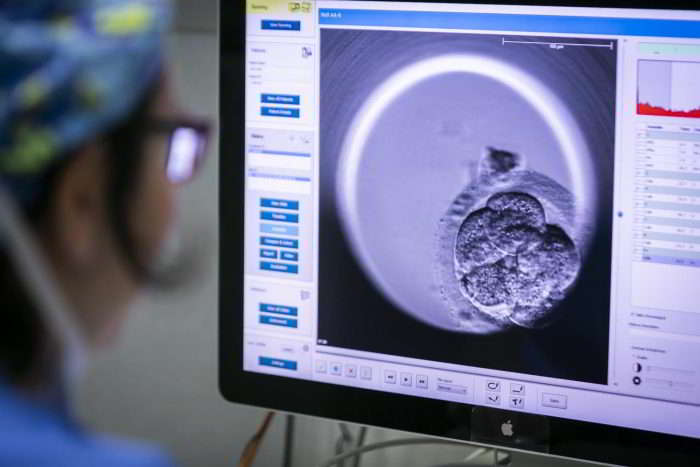

Once fertilised, the eggs are monitored in the laboratory as they begin to develop into embryos. In the following days, they will be graded based on their embryonic development, according to their morphology and ability to divide. PGT-A is performed at blastocyst stage, which is day 5 or 6 of development. A tiny sample of cells is extracted from the embryo using a needle, called a biopsy. The cells are taken from the trophectoderm, which will go on to form the placenta, so biopsy does not cause any harm to the embryo. Using NGS (Next Generation Sequencing), the number of chromosomes in the cells are examined to determine the genetic health of the biopsied embryos. During this time, the embryos are cryopreserved until the results are received.
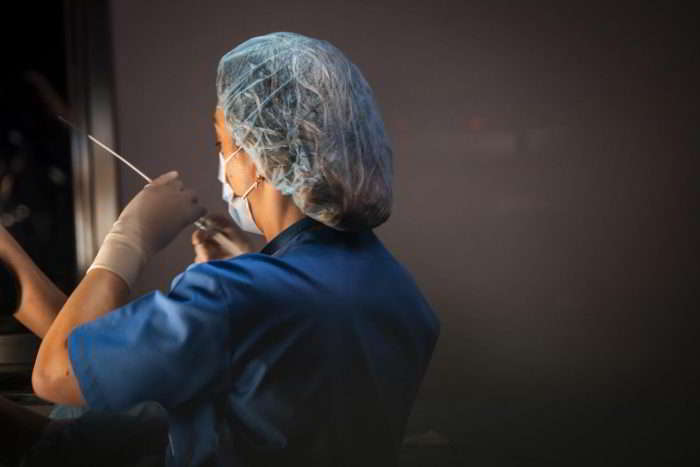
Once the healthiest embryo is selected, it is thawed safely and transferred into the uterus in an embryo transfer procedure. The procedure involves placing an embryo into the uterus using a narrow catheter. It is simple, quick and painless and is carried out in the clinic. After a few minutes rest, you can leave the clinic and carry on your day as normal.



In some cases, there are healthy embryos remaining after an IVF with PGT-A treatment has taken place. These embryos can be vitrified and preserved for a later cycle, removing the need to undergo ovarian stimulation, an egg collection and additional genetic screening.
Couples with repeat miscarriages, multiple IVF failures, previous pregnancy with chromosomopathies and advanced maternal age
Each type of PGT has its individual characteristics, but in all of them it is necessary to perform an in vitro fertilisation cycle in order to genetically analyse the resulting embryos. After 13-15 days of ovarian stimulation, the eggs are obtained (follicular puncture). Once they are fertilised with the donor’s or the partner’s sperm, the embryos are cultured until the blastocyst stage, which corresponds to days 5 or 6 after the puncture. At this point the embryo biopsy is performed and on the same day the embryos are frozen until the genetic results are obtained, which usually takes 10-15 days. In a subsequent cycle, and after endometrial preparation, the chromosomally normal embryos will be transferred.
PGT-A may increase the success rates of an IVF cycle, especially in infertile patients with a history of unsuccessful treatment, in cases of advanced maternal age, couples with repeated miscarriages/implantation failures, and in couples with a high risk of miscarriage.
Mainly by means of high-resolution genetic techniques such as NGS (next generation sequencing).